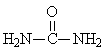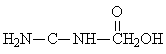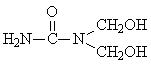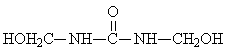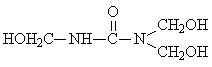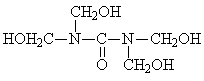Comparative Study of Synthesis Procedures for Urea - Formaldehyde Resins (Part I)
Mathew Obichukwu EDOGA
Chemical Engineering Department, Federal University of Technology, Minna, Nigeria, kovoabdulsalami@yahoo.com
Abstract
This paper presents initial results from modifying the conventional synthesis procedure of UF resins, that is reacting urea and formaldehyde first in alkaline medium and subsequently in mild acid medium at reflux (conventional two - stage alkaline - acid synthesis), by conducting the reaction only in purely acid medium at 50oC (modified one - stage acid synthesis).
In another attempt, the reaction was carried out in a neutral environment and at atmospheric temperature (modified one - stage neutral synthesis). The structural analysis of the various UF resin samples was carried out.
The results showed that the UF resins (II) synthesized in acidic media, particularly that of pH 1.0, showed features in their spectra which are consistent with a tri -, and/or tetra - substituted urea in the reaction to give a 3 - dimensional network UF resin. A 3 - dimensional network resin is characterized by little or no hydroxyl groups with increased cross-link-density and hydrophobicity and hence improved hydrolytic stability. For the resin (III) obtained from the conventional method, the only clear distinction between spectra for the resins (II) synthesized in acid media is the presence of diminished peaks for methylene groups at 2.2 ppm. The resins (I) synthesized in neutral medium exhibited almost the same structural characteristics as those of the resin samples (III) prepared conventionally. The result also showed that UF resin samples (I and III) possessed 5 - 17 percent of free formaldehyde; the UF resin sample (II) had approximately zero formaldehyde percentage.
Keywords
Urea-Formaldehyde resin, Synthesis- Procedures, Conventional method, Acid - modified procedure, Hydrolytic Stability.
Introduction
At present, the principal petrochemical adhesives often used in furniture industries are formaldehyde - condensation plastics.
These are urea - formaldehyde (UF), phenol - formaldehyde (PF), phenol - resorcinol - formaldehyde (PRF), and melamine - formaldehyde (MF), plastics. It is interestingly of note that bonding with UF adhesive is cheaper than bonding with PF adhesive, and it permits the formation of strong bonds under a wider variety of conditions. Moreover, since the manufacturing costs of urea - formaldehyde resins is relatively the least expensive synthetic organic resin as well as the most versatile in wood - adhesive bonding worldwide. However, the poor durability and stability of UF bonded products limit their use to interior, non - structural applications. These limitations are evidenced for example, in strength losses of UF bonded joints; irreversible swelling of UF bonded composite panels and formaldehyde release [[1]-[10]]. A significant improvement in the durability - stability of UF bonded wood products and drastic reduction in the formaldehyde emission would broaden the applications and markets for the products and thereby allow substitution of the UF - resin for more expensive, less versatile and more supply - dependent PF - system.
Several attempts have been made to develop hydrolytically stable and durable UF systems. For example, since formaldehyde - to - urea molar ratio (F/U) governs to some extents the stability and durability of wood joints [[11]] and particle board [[12], [13]], the effects of resin molar ratio from about 2.0 - 1.2, have been examined. Modification of the resins with several modifiers has also been studied [6, [14]-[17]]. The effectiveness of a variety of cross - linking agents has also been studied: trimethoxymethymelamine and dimethoxymethyl melamine were found to be beneficial for durability [[18]]. A remarkable improvement in stability - durability has been made by incorporating some suitable modifiers and buffers into the resins that are capable of neutralizing the acid - catalysts used as curing agents [14, [19], [20]].
Incorporating modifiers, cross - linking agents and other additives into UF resin, results in improved performance which is still inferior to that of PF -, or MF resin. This suggests that the optimum resin composition of UF resin has not been achieved. More so, it has been suggested that conventional concept of a 3 - dimensional infinite network may not always apply to cured UF resins [[21], [22]]. Traditionally, PF -, or MF resins are widely used in the manufacture of exterior - type wood products. However, phenol - formaldehyde resins currently the major adhesive family used in exterior - grade wood - adhesive bonding has recently experienced severe price hikes because of their dependence on petroleum as a raw material source. Petroleum is a finite natural resource subject to the vagaries of market forces. Improving the durability - stability of UF resins as a potential substitute for more petro-chemical dependent, less versatile, and more supply - sensitive PF resins, will be a welcome development for both manufacturer and users alike. The present study is one of such attempts.
The overall objective of the present study therefore was to improve the hydrolytic stability as well as reduce to the barest minimum the free formaldehyde content of the UF resins so as to widen their applications to exterior - grade structural uses. Specifically, the aim was to develop UF resins with increased hydrophobicity and crosslink density by initiating various synthesis procedures cum modifying the resins with ethylated urea ether resin.
Experimental Procedure
Materials and Method
The chemicals employed are described in Table 1. Throughout this paper, we used either the commercial names of the chemicals or our code names.
Conventional Two - Stage Alkaline - Acid Synthesis of UF Resin (UF III)
One hundred and thirty five cubic centimeters (135 cm3) of 37 - 40% aqueous formaldehyde solution whose pH had been adjusted in the range of 7.5 - 8.0 using 1 mole sodium hydroxides (2-5 cm3) was charged into a round bottom water jacketed glass reactor. Stirring was initiated using a mechanical stirrer. Fifty grams (50g) of urea was also charged into the reactor containing the formalin, and the mixture was stirred thoroughly. The reaction mixture was heated at reflux for 15 minutes after which the pH was then adjusted to 4.5 -5.0 with lactic acid. The mixture was refluxed again until the aliquots gave rise to a precipitate on addition to a large volume of water after which the pH was adjusted to 7.5-80 with 1 mole sodium hydroxide solution. The final solution (UF resin liquor - sample III) was concentrated by vacuum distillation until it attained a syrup like consistency.
Table 1 Experimental Materials
|
Material |
Source |
Comments |
|
Urea (U) (CO (NH2)2) |
BDH chemical, Poole, England |
mp 134oC Analytical grade |
|
Formaldehyde (F), (HCHO) |
BDH chemical, Poole, England |
37-41% aqueous Analytical reagent |
|
Urea - formaldehyde Concentrate (UF concentrate) |
National Fertilizer Company, Nigeria, NAFCON 85 Ltd. |
85% aqueous solution 15% H20 |
|
(CO (NH2)2) HCHO |
|
25% (NH2)2 Co, 60% CH20 density 1.33 gkm viscosity 50cp at 25oC |
|
Sulphuric acid (H2S04) |
BDH chemical, Poole, England |
98% concentrated Analytical grade |
|
Ethanol(C2 H50H) |
BDH chemical, Poole, England |
Anhydrous Analytical grade |
|
Monosodium dihydrogen Phosphate (NaH2Po4) |
BDH chemical, Poole, England |
Analytical grade |
|
Ammonium sulphate (NH4So4) |
BDH chemical, Poole, England |
Industrial grade |
|
Kaolin (Al2 Si2 O5 (OH)4) |
African Timber & Plywood, Sapele, Nigeria (ATP) |
Industrial grade |
Modified One - Stage Acid Synthesis (UF II)
One hundred cubic centimeter of urea formaldehyde concentrate whose F/U ratio was 4.80 at pH 8.5 was charged into a round - bottom water jacketed glass reactor. The pH of the concentrate was adjusted to 1.0 with a 50% H2S04 solutions, which caused the temperature to rise to 50oC. About 0.25g NaH2PO4 was used to maintain the pH at 1.0.
21.06 g of urea (0.35 moles) was slowly charged into the reaction mixture over a 15 minute time interval. The rate of addition was regulated, and the temperature was maintained at 50oC with the chiller thermocirculator. The viscosity built up was monitored at regular intervals of three minutes using brookfield synechro-lectric, until a target viscosity of 300cp was attained. At this viscosity, it was observed that the temperature and pH simultaneously dropped by 2oC and 0.1, respectively.
At the end of the 15 - 30 minutes, the F/U ratio was 2.94, and the pH of the reaction mixture was adjusted with a 50% NaOH solution, and 1.5cm3 of 100% triethanolamine to 6.5 and later to 7.5, respectively. 126.5g of urea (2.11 moles) was added to the reaction mixture which was further refluxed for about 15 minutes to ensure the completion of the reaction between urea and formaldehyde to form UF resin (II) sample.
The same experiment was repeated for pH 3.0 and pH 5.0, respectively. The synthesized resins at pH 1.0 and 50oC were modified by incorporating ethylated urea ether resin (EUER) during and after resin synthesis.
Modified One - Stage Neutral Synthesis (UF I)
A 185 cm3 volume of 37 - 40% aqueous formalin initially at a pH of 2.5 was adjusted to 8.5 with about 2.4 cm3 ammonia, 0.3 cm3 dilute hydrochloric acid, and 3.5 cm3 of 1M potassium hydroxide solution. The solution was added to a mixture of 90g of urea, 3g of hexamine and 2g of ammonium chloride, in a glass reactor to give a urea/ formaldehyde mole ratio of 1.2.2. The mixture was swirled with magnetic stirrer to dissolve and form a homogeneous solution at a pH of 8.2. The reaction mixture was carried out in absence of heat at room temperature for 12 hours.
Resins Characterization
NMR spectra of the various samples were analyzed with a Perkin Elmer varian Em 350L model, spectrometer. In each run, about 0.8mg of the synthesized UF resin was used. Deuterated chloroform/carbon tetrachloride was used as solvents. The instrument working parameters for the quantitative analyses of the samples were as follows: 10/20ppm sweep width, 2 minutes sweep time, 106 - 107 spectra amplitudes and 0.05 second filter, 0.05mg reference power and zero ppm end sweep.
Infrared Spectroscopy
Infrared spectra of the various resin samples in potassium bromide pellets were obtained using ATI Mattson Genesis Series Fourier Transform Infrared Spectrophotometer (FTIRTm). About 0.7mg of the powdered sample was mixed with 281mg of potassium bromide and was further ground in an agate mortar and pestle. The mixture was then transferred to a die and pressed into a disc/pellet.
The transmission spectrum was recorded over the range of 400 -500 cm-1.
Determination of Free Formaldehyde Content
Fifty cubic centimeters (50 cm3) of 1 mole of pure sodium sulfite solution was prepared in 500 cm3 - flask. Three drops of thymolphthalin indicator were added. The mixture was carefully neutralized by titration with 1N hydrochloric acid until the blue colour of the indicator disappeared.
Twenty-five grams of the resin sample at pH 7.1 was added to the sodium sulphate solution. The resulting mixture was titrated with 1N hydrochloric acid until complete decolorization was obtained. The experiment was carried out once at the end of the condensation reaction of urea and formaldehyde.
The free formaldehyde (%) content was calculated from the formula given below.
% CH2O = (Net (cm3) acid · Normality of acid · 3.0) ·100 / Weight of Resin sample.
Results and Discussion
NMR and IR Spectra
Figures 1 - 3 give the proton NMR spectra of the UF resins obtained at various synthesis procedures. The solution state spectrum of UF resin (I) synthesized at atmospheric conditions is given in Figure 1(a). The appearance of amine groups [-NHCH2NH2; 1.7 ppm] and primary amide hydrogen [- NHCONH2; 4.9 ppm], as well as the absence of methylene protons [-CH2: 2.2 - 2.5 ppm] indicate that there is absence of reactions due to polycondensation or methylenization. This is suggestive of polyaddition. The prominent appearance of peak at 3.4 ppm also shows clear evidence for the presence of methylol groups [-NHCH2OH; 3.4ppm].
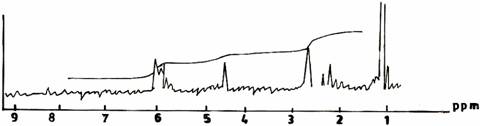
Figure 1(a).UF Resin I Synthesized Conventionally
The spectrum of the UF resin (II) prepared at F/U ratio 1.0, pH 1.0, and at a temperature of 500C (Figure 1(b), that is, by modified synthesis procedure differs from that of the UF resin (I) in four distinct regions. These are due to: (1) disappearance of the integral trace for the amine groups [-NHCH2NH2; 1.7 ppm]; (2) appearance of essentially equal amount of two different methylene hydrogens [2.3 - 2.5 ppm]; (3) diminished height and multiplicity of peaks around 3.8ppm with a shoulder at 3.5ppm which may be due possibly to a free OH group; (4) initiation of a small peak at 6.0ppm due to secondary amide hydrogens [-NHCONH2; 6.0 ppm]. A band at about 1.7 ppm (Figure 1(a) disappearing in Figure 1(b) on reacting urea and formaldehyde in acidic medium (pH 1.0), as well as its replacement with bands at 2.3 ppm and 2.5 ppm is consistent with the formation of methylene between secondary -, and/or tertiary amide hydrogens. Consequently, the diminished appearance of the peak at 5.0 - 4.8 ppm due to primary amide hydrogens in Figure 1(b) also shows that the UF structure became more cross linked due to the methylenization reaction.
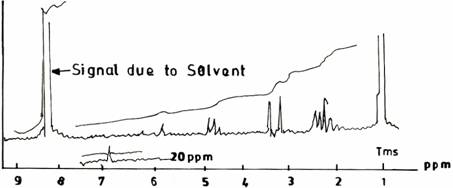
Figure 1(b). UF Resine II Synthesized at pH 1.0, F/U 1.0, 50°C
In general, the only clear distinction between spectra for modified reins (II), and the conventional UF resin (III) (Figure 1(c)) is the presence of diminished peaks for methylene groups at 2.2 ppm.
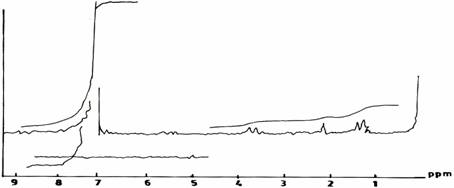
Figure 1(c). Commercial AT & P UF Resine III
On varying the pH values during the modified resin - synthesis (II) in acid environments, the spectra showed a similar range of resonance as did the spectrum of the control UF sample of Figure 1(b). The spectrum of UF resin sample (Figure 2(a)) synthesized at pH3.0, F/U 1.0 and at a temperature of 500C, showed a slight decrease in the height of peaks of the methylene protons. The solution state of UF resin sample prepared at pH 5.0, F/U 1.0, and at a temperature of 500C, gave a distinct specturm (Figure 2(b)) from the others, and it is difficult to interpret definitely.
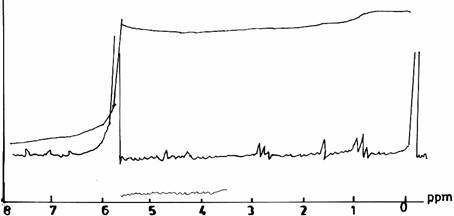
Figure 2(a). Proton NMR Spectra of UF Resins I, II, III at pH equal with 30
The spectra obtained from reacting urea and formaldehyde at F/U ratio 1.3 (Figure 3(a)) and F/U ratio 1.5 (Figure 3(b)), both at pH 1.0 and a temperature of 500C, possessed a similar range of resonance. Their spectra differ slightly from that of the UF resin (II) (Figure 1(b)) in the appearance of peaks at 5.7 ppm and 8.7 ppm, which may be due to the presence of ester group, and respectively the methylol group on secondary amide hydrogens.
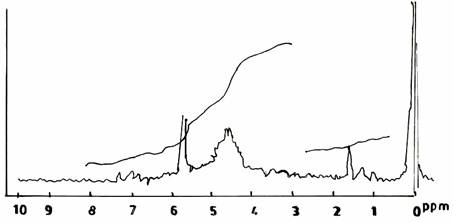
Figure 2(b). Proton NMR Spectra of UF Resins I, II, III at pH equal with 50
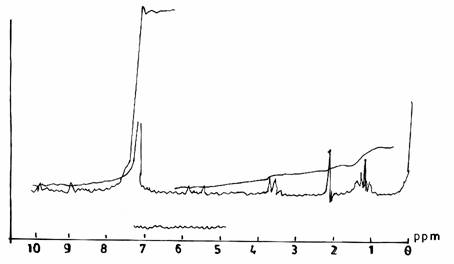
Figure 3(a). Proton NMR Spectra of UF Resins I, II, III at F/U mole ratio 1.3, pH 10, and 50°C
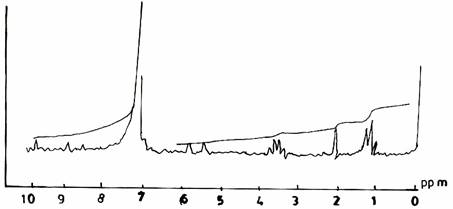
Figure 3(b). Proton NMR Spectra of UF Resins I, II, III at F/U mole ratio 1.5, pH 1.0, and 50°C
A summary of the possible products of the condensation reactions between urea and formaldehyde in slightly alkaline, neutral and purely acid (pH 1.0) conditions based on the integral traces of the proton NMR spectra cum the assignments of their chemical shifts has been given in Table 2.0. The structures of these products indicate that there are only three types of protons; the methylene, hydroxyl, and the amide, protons. Their NMR proton ratios are also given. The assignments of chemical shifts of the spectrum (Figure 1(b)) of the UF resin (II) prepared in pure acid medium of pH 1.0 are: multiple methylene proton [CH2; 2.5 - 2.2 ppm], free hydroxyl proton [-OH; 3.4ppm], as well as the amine groups [-NH2; 1.5 - 1.3 ppm], both from TMS. Their integral traces for that of UF resin spectrum synthesized conventionally (Figure 1(c)) is in the ratio of 2:1:1. The chemical shift calculated from shoolerys rule (John and Marijorie, 1977) [[23]], agrees with these assignments. Considering all the possible products and structures given in Table 2, the appearance of these signals - CH2-, OH, - NH2, in the ratios of 2:1:1 and 6:3:1, suggests that urea is di-, and tri- substituted in the products respectively. More so, the proton NMR of Figure 1(a), is suggestive of poly addition of urea and formaldehyde which invariably suggests that urea is mono - substituted.
The IR spectra of the same UF resin samples for which NMR speatra were recorded are shown in Figures 4-6.
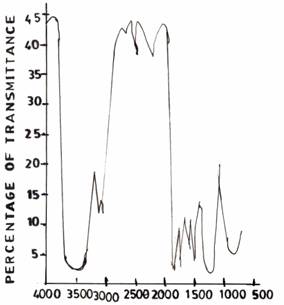
The spectrum of the UF resin (I) (Figure 4(a)) shows a strong absorption band at 3389 cm-1. This band is broad and typical of hydrogen bonded N-H and OH. However, when the reaction was conducted in acid medium (pH 1.0), the spectrum of the UF resin (II) (Figure 4(b)) in the 3360 - 3390 cm-1 region is sharper than that of UF resin (I) (Figure 4(a)) in 3389cm-1. One strong absorption is observed at 1610cm-1 which has been assigned to carbonyl C = 0 stretching vibration indicating the presence of tertiary amide. For the resin (III) synthesized conventionally, the spectrum (Figure 4(c)) differs from those of UF resins II and I in two distinct regions. These are due to the presence of a relatively broad peak at 3360 - 3390 cm-1 due to hydrogen bonding; and the appearance of sharp peak in the region of 2600 - 2190cm-1 due possibly to the presence of primary -, and/or secondary amides.
|
|
|
|
Figure 4(a). UF Resin I |
Figure 4(b). UF Resin II, pH 1.0, F/U 1.0 |
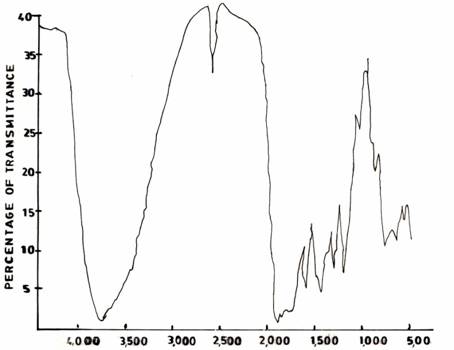
Figure 4(c). Commercial AT and P Resins (KAOL Modified)
|
|
|
|
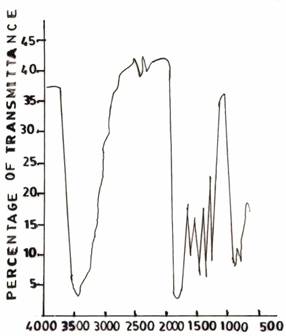
Figure 5(a). Infrared Adsorption Signals of UF Resins at pH 1.0, F/U 1.0, 50°C |
Figure 5(b). Infrared Adsorption Signals of UF Resins at pH 3.0, F/U 1.0, 50°C |
On varying the pH values pH 3.0 (Figure 4(a)) and pH 5.0 (Figure 5(b)), respectively, during resin synthesis, the band at about 3360 - 3390cm-1 got sharper compared to that of the control UF sample (Figure 1(b)). The sharpness of these bands many indicate a reduction in the extent of hydrogen bonded interaction which is expected as the structure becomes more cross-linked due to methylenization reaction.
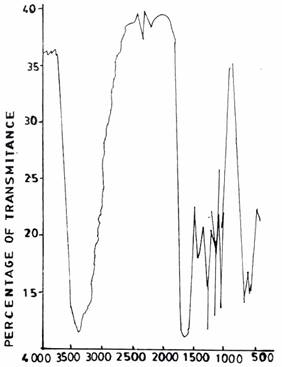
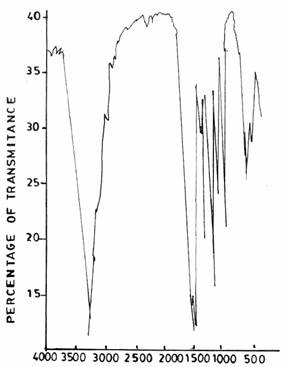
The spectra of UF resins prepared at F/U 1.3 (Figure 6(a)) and F/U 1.5 (Figure 6(b)), respectively, both at a temperature of 500C and pH 1.0 resemble those of UF resins prepared at pH 3.0 (Figure 5(a)) and pH 5.0 (Figure 5(b)).
The present IR spectra resemble those of cured UF resins obtained by Brown et al (1991) [[24]]. Consequently, the IR absorptions are supportive of the 1NMR assignments earlier made.
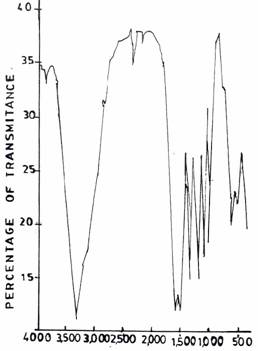
Figure 5(c). Infrared Adsorption Signals of UF Resins at pH 5.0, F/U 1.0, 50°C
The information obtained from the [[25]] NMR and IR spectra of the UF sample (II) (that is the one synthesized at pH 1.0, F/U 1.0 and at 500C) is consistent with structure (5) of Table 2, out of the six possible structures that may result from the polycondensation of urea with formaldehyde.
This observation is in agreement with Williams (1983) [18] postulation that employing modified one - stage acid synthesis would produce high cross-link density, and a 3 - dimensional infinite network.
The present finding, therefore, supports the earlier claim that synthesizing UF resin in purely acid environment (pH 1.0) will increase the probability of formaldehyde - substitution of the hydrogen atoms attached to urea to form either tri-, and/or tetramethylol ureas.
As a result of the above reasoning, the resins hydrophobicity and cross-linking ability would be enhanced significantly.
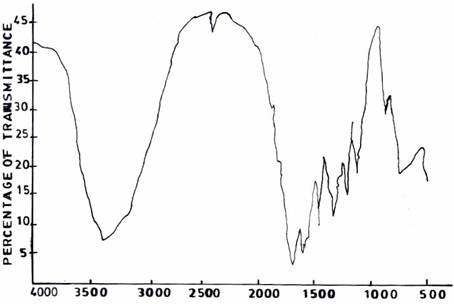
Figure 6(a). Infrared Adsorption Signals of UF Resins Synthesized at pH =1.0, F/U = 1.3, 50°C
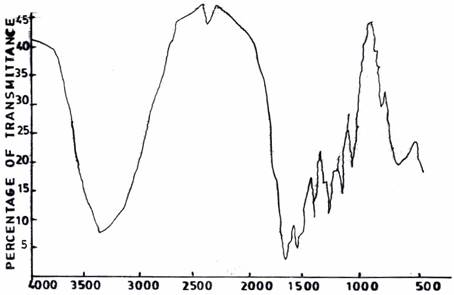
Figure 6(b). Infrared Adsorption Signals of UF Resins Synthesized at pH =1.0, F/U = 1.5, 50°C
Table 2. The possible products of UF Reactions
|
Material |
Structure |
Proton Ratio |
|
Unreacted material (Urea + formaldehyde) |
|
1 : 1: 1 |
|
Mono-methylolurea |
|
2: 1: 3 |
|
Di-methylolurea |
|
2: 1: 1 |
|
Di-methylolurea |
|
2: 1: 1 |
|
Tri-methylolurea |
|
6: 3: 1 |
|
Tetra-methylolurea |
|
2: 1: 0 |
Percentage Free Formaldehyde Content
Table 3 gives the percentage free formaldehyde content of the UF resin samples obtained from the three different UF resin synthesis procedures namely the conventional; the modified acid medium -, and the modified neutral medium, methods. For the conventional method, the percentage free formaldehyde content at the end of the reaction (45-50 minutes) is 17%; 5% for the modified neutral medium at the end of the reaction (21 hours); and approximately zero percent for the acid modified synthesis procedure at the end of the reaction of 35 - 50 minutes.
Table 3. Percentage Free Formaldehyde Content
|
Synthesis Procedure |
%CH2O Content |
|
Conventional method |
17 |
|
Modified neutral medium |
5 |
|
Acid modified method |
0 |
From Table 3, it can be clearly seen that the acid modified method, had approximately zero formaldehyde content implying that bonding with UF resins of the type would therefore emit no free formaldehyde.
As a consequence, indoor air pollution by formaldehyde issuing from commercial products containing urea - formaldehyde resins would be drastically reduced, and that makes UF resins environmentally friendly.
Overall, the modified acid - synthesis procedure produced UF resins with zero percent free formaldehyde content cum increased formaldehyde - substitution of the hydrogen atoms attached to urea to form either tri - and/or tetramethylolurea. This is a good indication that the resin would produce a three dimensional network cured UF resin and invariably possess greater hydrophobicity. By extrapolation, it would increase the cured resin strength, and hence improved stability-durability. On the other hand, the conventional and the modified neutral - synthesis procedures produced reins characteristic of mono - and/or di-methylolurea (Table 2) and of 5 - 17 percent free formaldehyde content. This later trend increases the international concern pertaining the application of UF resins in wood - products manufacture since the indoor air pollution by formaldehyde issuing from the commercial products containing urea - formaldehyde (UF) resins has been an important concern of wood product industry in many countries for at least 20 years and particularly for the last 12 years of so (Myers, 1988).
Conclusions
The overall conclusions emerging from the synthesis of UF resins at various synthesis procedures are that:
1. NMR and infrared spectra of UF resins show differences in the chemical structure of conventionally synthesized UF resin; acid - modified - synthesized UF resin as well as the neutral modified - synthesized resin.
2. For acid - modified resin synthesis procedure, a resin characteristic of tri - and/or tetramethylolurea was obtained which invariably possessed approximately zero percent free formaldehyde content. Consequently, the zero formaldehyde content makes the final wood products bonded with the resin environmentally friendly. The resin has the potential of producing a three dimensional network if properly cured and hence greater hydrophobicity and cross linking.
References