Corrosion Behavior of Austenitic and Duplex Stainless Steels in Lithium Bromide
Ayo Samuel AFOLABI*, K. K. ALANEME, and Samson Oluwaseyi BADA
School of Chemical and Metallurgical Engineering, Faculty of Engineering and the Built Environment, University of the Witwatersrand, P/Bag X3 Wits 2050 Johannesburg, South Africa.
*Correspondence to: A. S. Tel. +27 11 717 7539; Fax: +27 11 717 7591.
E-mail: afolabisammy@yahoo.com
Abstract
The corrosion behavior of austenitic and duplex stainless steels in various concentrations of lithium, bromide solution was investigated by using the conventional weight loss measurement method. The results obtained show that corrosion of these steels occurred due to the aggressive bromide ion in the medium. Duplex stainless steel shows a greater resistance to corrosion than austenitic stainless steel in the medium. This was attributed to equal volume proportion of ferrite and austenite in the structure of duplex stainless steel coupled with higher content of chromium in its composition. Both steels produced electrochemical noise at increased concentrations of lithium bromide due to continuous film breakdown and repair caused by reduction in medium concentration by the alkaline corrosion product while surface passivity observed in duplex stainless steel is attributed to film stability on this steel.
Keywords
ASS, DSS, lithium bromide, corrosion, surface passivity
Introduction
Ever since the discovery of stainless steels in 1913 [1], several groups of these steels have emerged. Among these groups are martensitic, ferritic and austenitic stainless steels (ASS). This classification is based on their chemical composition, metallurgical structure and mechanical properties [2]. However, the search for improved material to meet the challenges of ever dynamic technological world has led to recent discovery of duplex stainless steel (DSS). The corrosion behavior of these steels in a particular medium is determined by their passive nature [3,4], alloy composition [5,6] and precipitation morphology [7,8]. The intactness of passive film on these metals is dependent on its stability in the medium of exposure.
DSS has a dual phase microstructure of approximately equal volume fractions of ferrite and austenite [9]. This steel possesses a favorable combination of mechanical strength and corrosion resistance in chloride ion-containing environments [10] and it is widely used in industrial sectors such as petrochemical, power generation and transport [9]. It has the ability to accommodate higher chromium and molybdenum contents than many other stainless steels. These alloying elements, as well as nickel and nitrogen, are known to enhance the resistance to pitting corrosion, crevice corrosion and chloride stress corrosion cracking [9].
Corrosion occurs in many forms in structures made of these steels during service in various media. Some of these forms are intergranular [10,11], pitting, sulfide stress cracking, chloride stress cracking [12,15] and stress corrosion cracking [16,17]. Weld decay is also common in sensitized stainless steel structures [18,19]. The consequences of these forms of corrosion are obvious; varying from enormous material losses to unreliability of operating equipment which may lead to catastrophic consequences involving both men and money.
Refrigeration technology extensively utilizes lithium bromide-based brines as working fluids. Concentrated solutions of this salt have desirable properties as absorbents because of their high hydration heat, high solubility of solid phases, good thermal stability, and appropriate viscosity [20]. However, lithium bromide solutions can cause serious corrosion problems in metallic components in both refrigeration systems and heat exchangers in absorption plants [21]. In particular, corrosion problems are exacerbated by the recent trend toward using triple effect technology, which involves the use of higher temperatures and more corrosive salts in addition to lithium bromide [20]. Thus this work is aimed at investigating and comparing the corrosion behaviour of austenitic and duplex stainless steels in various concentrations of this medium.
Experimental
A 15mm diameter austenitic stainless and an 11mm duplex stainless steels were used in this work. They were obtained from the Universal Steel Limited, Ikeja, Lagos. Their chemical compositions as supplied by the manufacturer are as shown in Table 1.
Table 1: Chemical compositions (wt %) of ASS and DSS samples
|
Composition |
C |
Si |
S |
P |
Mn |
Ni |
Cr |
Mo |
Cu |
Fe |
|
ASS |
0.085 |
0.486 |
0.015 |
0.039 |
1.296 |
11.737 |
19.146 |
0.078 |
0.207 |
Bal. |
|
DSS |
0.03 |
0.8 |
0.02 |
0.03 |
1.5 |
6.5 |
21-23 |
0.082 |
0.5 |
Bal. |
Corrosion samples of these steels were prepared using the procedure and precautions described by [2224]. The samples were totally immersed in different concentrations (0.25M, 0.5M, 0.75M, 1.0M, 1.25M and 1.5M) of lithium bromide solution. The variation in the lithium bromide concentration is to evaluate the influence of mild and aggressive concentrations of the solution on the corrosion behavior of the austenitic and duplex stainless steels. The corrosion behavior of the steels was assessed using the conventional weight loss measurement at every five day interval for a total period of forty days using a digital weighing balance.
Results and Discussion
Figures 1-6 show the comparative plots of weight loss versus exposure time for AAS and DSS immersed in different concentrations (0.25M, 0.5M, 0.75M, 1.0M, 1.25M and 1.5M) of lithium bromide solution. It can be seen from these Figures that for all concentrations of lithium bromide, the DSS show less susceptibility to corrosion in comparison to the ASS. This is attributed to the matrix structure of the duplex stainless steel, which consists of ferrite and austenite. This composite structure does not show adverse tendency for galvanic corrosion due to the fact that the two phases that make up the duplex structure are of almost equal volume fraction [25]. Thus the requirement of small anode-large cathode for intense corrosion is not fulfilled [26]. Also the ferritic phase is noble due to the fact that it contains a small amount of carbon atoms interstitially dissolved in it (maximum 0.02 wt% C), this makes it less corrosion susceptible in comparison to the austenitic phase which has a greater proportion of carbon addition [27]. The composition of the DSS also shows that it has a higher proportion of chromium (21 23 wt% Cr) in comparison to the ASS (19.14 wt % Cr). The higher chromium percentage in the duplex structure makes the ferritic phase very stable and less prone to dissolution. It also contributes in increasing the overall corrosion resistance of the DSS due to the higher chromium oxide which potentially may be formed by it.
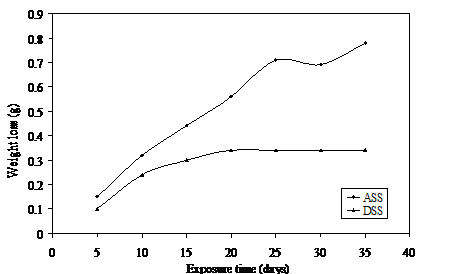
Figure 1. Weight loss versus exposure time for ASS and DSS immersed in 0.25 LiBr
Figure 1 shows the corrosion behaviour of the ASS and DSS samples in 0.25 M solution of lithium bromide. The Figure reveals a progressive weight loss in the ASS throughout the exposure time. This could be attributed to the aggressive bromide ion which continuously breaks down the protective film on this metal. Protective chromium oxide formed on the surface of the ASS in the presence of aerated concentration of lithium bromide could have been too weak or thin to prevent further penetration of bromide ion which is known for its aggressiveness and depassivation effect which usually lead to pitting [28]. DSS on the other hand shows a weight loss until the twentieth day of exposure while surface passivation of this steel is observed for the rest of the exposure period. The higher content of chromium in the composition of this metal is suspected to have produced a stronger and thicker chromium oxide protection to it but took a period of twenty days to stabilize this protective film on this metal.
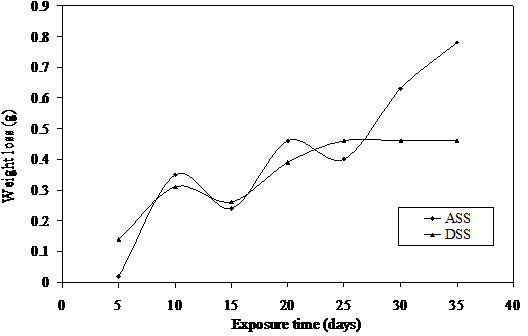
Figure 2. Weight loss versus exposure time for ASS and DSS immersed in 0.5M LiBr
In Figure 2, the weight losses of the two steel samples fluctuate up till twenty fifth day of exposure while ASS shows increase in corrosion after this period and surface passivation is observed for the rest of the exposure time in DSS. Similar features are observable in Figures 3 and 4 with the exception that surface passivation is really noted in DSS.
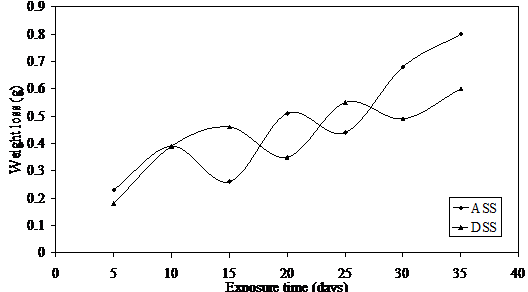
Figure 3. Weight loss versus exposure time for ASS and DSS immersed in 0.75M LiBr
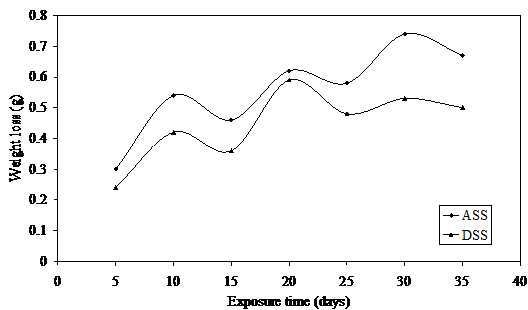
Figure 4. Weight loss versus exposure time for ASS and DSS in 1.0M LiBr
The weight loss fluctuations can be attributed to constant damage on the protective film by the notorious bromide ion and subsequent repair by the constantly formed protective chromium oxide on the surfaces of these metals. This phenomenon which could be referred to as electrochemical noise [29 31] is possible at increasing concentrations of this medium due to the presence of alkaline corrosion product which reduces the concentration of the lithium bromide medium.
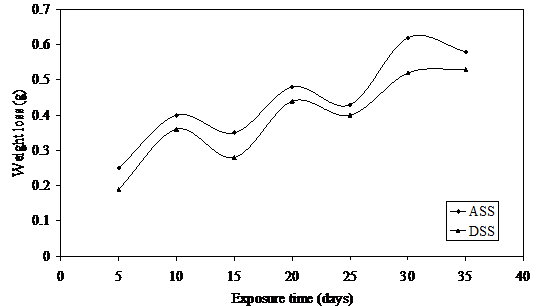
Figure 5. Weight loss versus exposure time for ASS and DSS immersed in 1.25M LiBr

Figure 6. Weight loss versus exposure time for ASS and DSS immersed in 1.5M LiBr
The intensity of this noise appears to increase in Figures 5 and 6 while slight passivation is noted in the last five days of exposure in DSS. Critical observation of these Figures reveals a slight drop in average weight loss values for both steels as compared to much lower concentrations of lithium bromide observed in Figures 1 and 2. Possible explanation for this occurrence is traceable to reduction in concentration of this medium with exposure time which has been previously discussed.
Conclusions
The following conclusions can be drawn from the studies conducted on electrochemical corrosion behavior of ASS and DSS in different concentrations of lithium bromide.
1. Corrosion of ASS and DSS occurred in lithium bromide due to the aggressiveness of bromide ion in the medium.
2. Corrosion susceptibility is more pronounced in ASS than DSS throughout the exposure time period studied. This is due to higher chromium content and equal volume proportion of ferrite and austenite in the structures of the latter.
3. Both stainless steels produced electrochemical noise at increased concentrations of lithium bromide due to continuous film breakdown and repair caused by reduction in medium concentration by the alkaline corrosion product.
4. Surface passivation observed only in DSS is attributed to film stability on this steel.
References
1. Bailey, F. W. J, Fundamentals of Engineering Metallurgy, Casell and Macmillan Publishers Ltd. New York, p. 77-86, 1972.
2. Dorsey, R. F Amstutz, R. W; Bruno, T. V; Doremusi, G. L Flamang, R. L. gerner, R. V; Roberson, G. R and Simmions, E. J, Corrosion control in petroleum products. TPC Publication No 5, NACE Publication, Houston Texas, 1979.
3. Okamoto, G. and Shibata, T, Passivity and breakdown of passivity of stainless steel, In Frankenthal, R. P. and Kruger, J. (Eds), Passivity of metals, Electrochemical Society Inc., New York, p. 646, 1978.
4. McBee, C. L. and Kruger, J, Nature of passive films on iron chromium alloys, Electrochimica Acta 17, p. 1337, 1972.
5. Cihal, V, Intergranular corrosion of steels and alloys, Elservier Amsterdam, p. 60, 1984.
6. Sedrick, A. J, Corrosion of stainless steels, John Wiley & Sons. New York, 1979.
7. Okamoto, G, Passive film of 188 stainless steel structure and its function, Corrosion Science 13, p. 471, 1973.
8. Foley, C. L., Kruger, J. and Bechtoldt, C. J, Electron diffraction studies of active, passive and transpassive oxide films formed on iron, Journal of Electrochemical Society 114, p. 994, 1967.
9. Sun, Z., Kuo, M., Annergren, I. and Pan, D, Effect of dual tourch on duplex stainless steel welds, Materials Science and Engineering A356, p.274282, 2003.
10. Wilson A, Stainless Steel Engineering, 2nd ed. Johnson Press London, , p. 45-60. 1995
11. Zheng, S., Shibata, T. and Haruna, T, Inhibition effect of metal cations to intergranular stress corrosion cracking of sensitized Type 304 stainless steel, Corrosion Science 47, , p. 10491061, 2005.
12. Aydogdu, G. H. and Aydinol, M. K, Determination of susceptibility to intergranular corrosion and electrochemical reactivation behaviour of AISI 316L type stainless steel, Corrosion Science 48, p. 3565 3583, 2006.
13. Pohjanne, P, Corrosion behaviour of austenitic stainless steels in chloride containing ozone solutions, Corrosion 376, p. 1 14, 1997.
14. Bellanger, G, Inhibition of localized corrosion for stainless steels in lowlevel radioactive water containing chloride, Corrosion Science 48, , p. 1379 1403, 2006.
15. Chen, Y. Y., Chou, L. B. and Shih, H. C, Factors affecting the electrochemical behaviour and stress corrosion cracking of Alloy 690 in chloride environments, Materials Chemistry and Physics 97, p. 37 49, 2006.
16. Okpala, A. N. and Jombo, P. P, Corrosion behaviour of stainless steel in seawater before and after welding. Nigeria Journal of Engineering Research and Development 3, (3), p 57, 2004.
17. Nishimura, R, Characterization and perspective of stress corrosion cracking of austenitic stainless steels (type 304 and type 316) in acid solutions using constant load method, Corrosion Science, Article in press, 2006.
18. Walker, P. M. B, Chambers materials science and technology dictionary, Chamber Harrap Publishers Ltd. Edinburgh, 1993.
19. Parvathavarthini, N., Dayal, R. K., Khatak, H. S. Shankar, V. and Shanmugam, V, Sensitization behaviour of modified 316N and 316L stainless steel weld metals after complex annealing and stress relieving cycles, Journal of Nuclear Materials 355, p. 68 82, 2006.
20. Herold, K.E. Radermacher, R. Klein, S. A, Absorption chillers and heat pumps, Boca Raton, FL: CRC Press, p. 101, 1996.
21. Garcia Anton, J. Perez Herranz, V., Guinon, J. L. and Lascoste, G, Use of differential pulse polarography to study corrosion of galvanized steel in aqueous lithium bromide solution, Corrosion 50, (2), p. 9197, 1994.
22. Chen, X. H., Chen, C. S., Xiao, H. N., Cheng, F. Q., Zhang, G. and Yi, G. J, Corrosion behaviour of carbon nanotubes-Ni composite coating, Journal of Surface and Coating Technology 191, p. 351- 356, 2005.
23. Ashassi-Sorkhabi, H. Ghalebsaz-Jeddi, N., Hashemzadeh, F. and Jahani, H, Corrosion inhibition of carbon steel in hydrochloric acid by some polyethylene glycols, Electrochimica Acta 51, p. 3848 3854, 2006.
24. Jabeera, B. Shibli, S. M. A. and Anirudhan, T. S, Synergistic inhibitive effect of tartarate and tunstate in preventing steel corrosion in aqueous media, Journal of Surface Science 252, p.35203524, 2006.
25. Martin, J. W. Doherty, R. D. and Cantor, B, Stability of microstructure in metallic systems, 2nd ed. Cambridge University Press, 1997.
26. Barnarjee, S. M, An introduction to science of corrosion and its inhibition, Oxoran Press. PUT Ltd, 1985.
27. Kamma, C. M, Design of microstructures by thermo mechanical treatment, Proceedings of the Nigerian Metallurgical Society, Kaduna, p. 8-18,1992.
28. Refaey, S. A. M, Taha, F. and ElMalak, A. M, Corrosion and inhibition of stainless stainless steel pitting corrosion in alkaline medium and the effect of Cl and Br anions, Applied Surface Science 242, p. 114120, 2005.
29. Millard, S. G, Reinforced concrete resistivity measurement techniques, Proceedings of the institute of civil Engineers 2, p. 7188, 1991.
30. Cottis, R. A., AlAnsari, M. A. and Pettiti, A, Electrochemical noise measurements for corrosion studies, Materials Science Forum 8, p. 741, 1997.
31. Afolabi, A. S. and Fasuba, O. A, Corrosion of steel reinforcements in concrete immersed in different environments, Nigerian Society of Engineers Technical Transcaction. 41, (4), p. 51 60, 2006