Morphology and Hardness Improvement of Lead Bearing Alloy through Composite Production: 75Pb-15Sb-10Sn/ 15% V/V SiO2 Particulate Composite
Linus Okon ASUQUO1, Aondona Paul IHOM1*, and Kunle MAYALEEKE2
1 Departament of Mechanical Engineering, University of Uyo, Uyo, Akwa Ibom State, Nigeria
2 Department of Metallurgy, National Metallurgical Development Centre, Jos, Plateau State, Nigeria
E-mails: draondonaphilip@gmail.com ; loasuquo@yahoo.com *; kmayaleeke@yahoo.com Corresponding author: Phone: +234-8035813571
Abstract
The morphology and hardness improvement of lead bearing alloy through composite production: 75Pb-15Sb-10Sn/ 15%v/v SiO2 particulate composite, was studied. 75Pb-15Sb-10Sn white bearing alloy produced at the foundry shop of National Metallurgical Development Centre Jos was used for the production of the composite using stir-cast method. The reinforcing agent was 63 microns passing particles of silica. This was produced from pulverizing quartz using laboratory ball mill. The specimens of the composite produced were then subjected to metallographic to study the morphology of the structures produced both in the as cast and aged conditions of the composite. The samples were also tested for hardness and the result showed that the as cast composite had a hardness value of 33 HRB which is an improvement over the hardness value of 27.7 HRB for the 75Pb-15Sb-10Sn alloy which was used for the production of the composite. The effect of age hardening on the produced composite was also investigated; the result showed that the maximum hardness of 34 HRB was obtained after ageing for 3 hours. The micrographs revealed inter-metallic compound SbSn, eutectic of two solid solutions-one tin-rich and the other lead-rich, reinforcing particles, and solid solution of β. The results revealed that particle hardening can be used to improve the hardness of 75Pb-15Sb-10Sn white bearing alloy for use as heavy duty bearing material.
Keywords
Morphology; Hardness improvement; Lead bearing; Alloy; Composite; Particulate.
Introduction
“White” bearing metals are either tin-base or lead –base. White bearing metals contain as much as 90% tin and up to 10% antimony. These two metals form “cuboids” crystals of the inter-metallic compound SbSn which are easily identified in the microstructure and which constitute the hard, low-friction phase. In lead-free white metals the cuboids are held in a matrix of the solid solution α whilst in white metals to which lead has been added in amounts up to 80% in order to reduce cost, the SbSn cuboids will be in a matrix consisting of a eutectic of two solid solutions-one tin –rich and the other lead-rich. SbSn inter-metallic compound has the tendency of segregation during solidification [1]. Lead base alloys possess low tensile strength of the order of 600 to 1000kg/cm2. Lead base alloys are costlier than zinc base alloys. Lead base alloys being toxic need careful handling. There is a gradual shift from the use of lead based alloy to lead free alloys [2], be it as the case may be a lot of lead bearing alloys are still used in some countries. It is also important to note that every research is important if it is able to solve a problem or to meet a need. A common die-casting lead base alloy contains 15% antimony, 5% tin, 0.5% copper, and the remaining lead. Antimony increases hardness whereas tin improves hardness, strength and fluidity of lead base alloys. Applications include accumulator plates, and terminal caps, radiation (x-ray) shielding, battery parts and light duty bearings [1].
Lead and tin alloys are used as bearing materials for antifriction bearings. When antimony is added, they are known as babbit metals. A typical composition of a lead based alloy is 75%Pb, 15%Sb and 10% Sn. Lead based alloys are softer and brittle than the tin base alloys. They have a higher coefficient of friction as compared to tin based alloys. Lead based alloys are suitable for light and medium loads whereas tin base alloys are preferred for higher loads and speeds. Lead base alloys are used in rail road freight cars. Lead base alloys have a solidus temperature of approximately 240°C. It has good ability to embed dirt, conformability to journal, corrosion resistance and very good seizure resistance, etc. [1,2]. Despite these properties the low hardness and strength of lead base alloys limit its use as a bearing material to light and medium loads bearings. There is need to improve the hardness of lead base alloys so that it can be use for higher loads and speeds. This can be done through composite production using the alloy as a matrix. The alloy will be reinforced with particulates to increase both strength and hardness [1,2]. According to Ihom et al. [3] composites combine the attractive properties of the other classes of materials while avoiding some of their drawbacks. They are light, stiff, and strong, and they can be tough. Metal matrix composites (MMCs) reinforced with ceramics or metallic particles are widely used due to their high specific modulus, strength, hardness and wear resistance. MMCs have been considered as an alternative to monolithic metallic materials or conventional alloys in a number of specialized applications. The majority of such materials are metallic matrices reinforced with high strength, high modulus and often brittle second phase in the form of fibre, particulate, whiskers, embedded in a ductile metal matrix. Particulate composites depend for their strength upon a degree of particulate hardening such materials include “cermets” which are a mixture of metal and ceramic substances and are generally compounded with the object of producing a combination of hardness and toughness such as would be required in a tool material. Yet a further group of composite materials relies upon dispersion hardening. Here the movement of dislocations is impeded by strong particles of microscopically dimensions only. The nature of the interface between the particles and the matrix has a bearing on the extent to which the load will be transferred from the matrix to the strengthening material [1,4].
Quartz, second most common of all minerals, composed of silicon dioxide, or silica, SiO2. It is distributed all over the world as a constituent of rocks and in the form of pure deposits. It is an essential constituent of igneous rocks such as granite, rhyolite, and pegmatite, which contain an excess of silica. In metamorphic rocks, it is a major constituent of the various forms of gneiss and schist; the metamorphic rock quartzite is composed almost entirely of quartz. Quartz crystallizes in the rhombohedra system. The size of the crystals varies from specimens weighing a metric ton to minute particles that sparkle in rock surfaces. Quartz is also common in massive forms, which contain particles ranging in size from coarse-grained to cryptocrystalline (grains invisible to the naked eye but observable under a microscope). The mineral has a hardness of 7 and specific gravity of 2.65. The luster in some specimens is vitreous; in others it is greasy or splendent (shining glossily). Some specimens are transparent; others are translucent. In the pure form, the mineral is colourless, but it is commonly collared by impurities. Quartz crystals undergo structural transformations when heated. Ordinary, or low, quartz, when heated to 573°C (1063.4°F), is converted into high quartz, which has a different crystal structure and different physical properties. When cooled, however, high quartz reverts to low quartz. Between 870° and 1470°C (1598° and 2678°F), quartz exists in the form called tridymite, and above 1470°C (2678°F), the stable form is known as cristobalite. At about 1710°C (3110°F), the mineral melts [4]. Deposits of different varieties of quartz abound in Jos-Nigeria and the clear transparent and lustrous type was selected for this research work. Based on the properties outlined above the material meets the specification expected of a reinforcing agent [5].
Bearing supports moving parts such as shafts, and spindles, of a machine or mechanism. Bearings may be classified as rolling contact and plain bearings. Giving an overview of bearing metals by several authors [1,6] the authors argued that the mechanical requirements of a bearing metal can only be met by the intelligent use of alloying. Some authors [1,2,7,8] argue that it can also be met through composite material. They all however, agree that a bearing must be hard and wear-resistant with a low coefficient of friction but at the same time be tough, shock resistant and sufficiently ductile to allow for “running in”. These properties of hardness, toughness and ductility cannot be found to the required degree in a single-phase alloy. Thus, inter-metallic compounds are hard and have a low coefficient of friction but are extremely brittle, whilst pure metals and solid solutions though ductile are usually soft and with a relatively high coefficient of friction. A suitable combination of mechanical properties can, however, is obtained by using an alloy in which particles of a hard inter-metallic compounds are embedded in a matrix of ductile solid solution or, in some cases a eutectic of two solid solutions. Alternatively pure metals or alloys can be used to produce composites with hard particulates embedded in the matrix of the metals and alloys. Pb-Sb-Sn alloy form inter-metallic compound SbSn in the matrix of the eutectic along side with β solid solutions. In a similar work carried out by Ihom [9] the author was able to show that the composite had increased hardness in the as cast state and also had increased hardness during three hours of ageing. The work revealed the morphology of the microstructure with inter-metallic precipitates which improved the hardness of the composite as well as the bearing properties of the Pb-Sb-Sn matrix composite. Khana [2] in his work found out that during the “running in” process the soft matrix tends to wear leaving the hard particles standing proud. This not only reduces the overall coefficient of friction of the bearing surface but also provides channels through which lubricant can flow [2,7-9].
The aim of this paper was to use the composite route to improve on the hardness characteristic of 75Pb-15Sb-10Sn bearing alloy and also to study the emerging morphology of the composite, so that it can be used for heavy duty loading and high speed devices.
Material and Method
The materials used for the work were 75Pb-15Sb-10Sn alloy with a hardness value of 27.7 HRB produced in the foundry shop of National Metallurgical Development Centre Jos, pure quartz from Jos deposit at Rukuba, clay, alumina, and silicon carbide powder, and water. The equipment used included, melting crucible furnace, heating oven, specimen lathe, Rockwell hardness tester, computerized metallurgical microscope, grinding and polishing disc, stirring device, electronic weighing balance, permanent mould, tongs, and quenching bath.
Composite Production
The composite was produced using 75Pb-15Sb-10Sn alloy with a hardness value of 27.7 HRB produced in the foundry shop of NMDC Jos. The charge to produce the composite was calculated based on the dimension of the test sample which was 20cmxØ2cm. The charge was introduced into the crucible furnace which was equipped with a mechanical stirrer. The furnace which was earlier on preheated to 300°C had the temperature raised to 500°C after charging. When the alloy completely melted, 15%v/v of quartz (SiO2) which was pulverized using laboratory ball mill to 63 microns passing was introduced into the melt. The mechanical stirrer was inserted and the stirring was done at the rate of 315rpm for 1 minute, to avoid freezing and the temperature was equally raised to 600°C. The melt was then quickly poured into permanent moulds which were sealed at the bottom and by the side with clay to avoid leakage. After cooling the solidified bars were removed; three test bars were produced. Lead is poisonous therefore all the workers wore nose mask to avoid inhaling the lead vapour.
Ageing
The bars were prepared into test specimens of 2cmx2cm using specimen lathe. Some of the specimens were heated to 300°C in the oven and held for 1 hour and then quenched in warm water. The specimens were then artificially aged in the oven at 100°C, for time ranging 1 hour to 4 hours. They were then subjected to hardness test and micro-structural examination.
Microstructure Examinations
The specimens were ground and polished. A belted grinding machine with grits 240-600 was used. The specimens were then transferred to a pre-polishing disc where alumina powder paste of 1 micron was used for pre-polishing. The specimens were finally polished on the finishing disc; 0.5 micron of alumina paste was used. It was ensured that the surfaces were devoid of scratches and it was thoroughly washed and dried using a hand blower to avoid chemical corrosion. 2% nital solution was used to etch the specimen and rinsing was done using clean water. It was then dried using a blower before transferring to the microscope for viewing and taking of the photomicrograph of the microstructure.
Results and Discussion
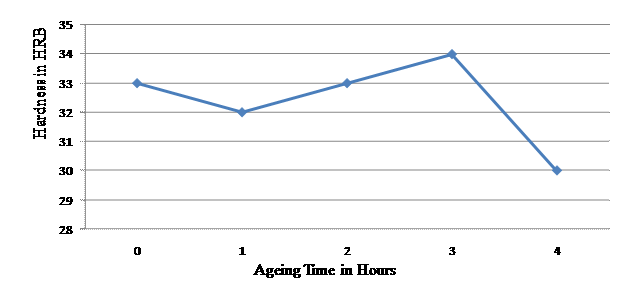
Table 1 shows the hardness values of the as cast composite and that of the composites age hardened at various ageing temperatures. The alloy matrix used for the production of the composite had a hardness value of 27.7 HRB. The as cast composite had a hardness value of 33 HRB, after ageing for 1 hour the hardness value dropped to 32 HRB. It however, rose to 33 HRB after ageing for 2hours and then 34 HRB after 3hours of ageing. The hardness then dropped to 30 HRB after 4hours of ageing.
Table 1. Hardness Values of 75Pb-15Sb-10Sn / 15%v/v SiO2 Particulate Composite in HRB
|
S/NO |
Composition |
Ageing Time in Hours |
Hardness Values in HRB |
|
1 |
75Pb-15Sb-10Sn/ 15%v/v SiO2 |
0 |
33.0 |
|
2 |
1 |
32.0 |
|
|
3 |
2 |
33.0 |
|
|
4 |
3 |
34.0 |
|
|
5 |
4 |
30.0 |
This drop in the hardness value can be traced to segregation effect which can be seen in the microstructure of the composite aged hardened for 4hours [7-8]. Another phase different from the eutectic matrix and the inter-metallic compound SbSn can be seen stretching through the microstructure. It is clear from this result that the age hardening of the composite did not result in reasonable increase in the hardness of the composite. This may be because no further phase precipitation or little occurred during the ageing process. The alloy matrix as indicated in research carried out by several authors [1-2,9-11] has precipitates of SnSb inter-metallic compound this is responsible for the hardness of the Pb/Sb/Sn alloy. The hardness of the as cast composite increased above that of the alloy matrix indicating that the silica reinforcement used was responsible for the increase in hardness of the composite over that of the alloy used as matrix. According to Taylor [12], the physical barriers in the form of a precipitate and reinforcement particles is provided to hinder the motion of the dislocation in the matrix of the alloy which leads to increase in strength and hardness. This can also be explained in terms of solid solution formation with the matrix and dispersion effect. Khanna [2] and Higgin [1] have agreed that metals and metal alloys may be strengthened and hardened by the uniform dispersed of several volume per cent of fine particles of a very hard and inert material. The mechanical properties are enhanced with increasing particulate content. This result also agrees with the result of several works carried out by different authors [13-16].
Figure 1 shows the effect of ageing time on the Hardness Values of 75Pb-15Sb-10Sn/ 15% v/v SiO2 Particulate Composite. The figure shows that as the ageing time is increased to 1 hr the hardness value dropped from 33 HRB to 32 HRB. The hardness then increased reaching a peak of 34 HRB at 3hours of ageing and then dropped to 30 HRB after age hardening for 4 hours. The hardness variation with respect to ageing time can be attributed to reinforcing particulate and precipitate distribution during the ageing process [1,13-16]. The result of the age hardening process has shown that the best ageing time for better result is 3 hours of ageing.
|
|
|
Figure 1. Effect of Ageing Time on the Hardness Values of 75Pb-15Sb-10Sn / 15% v/v SiO2 Particulate Composite |
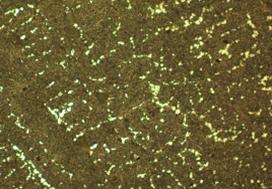
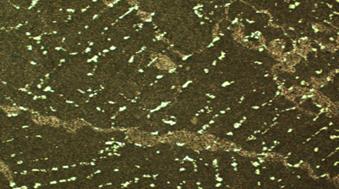
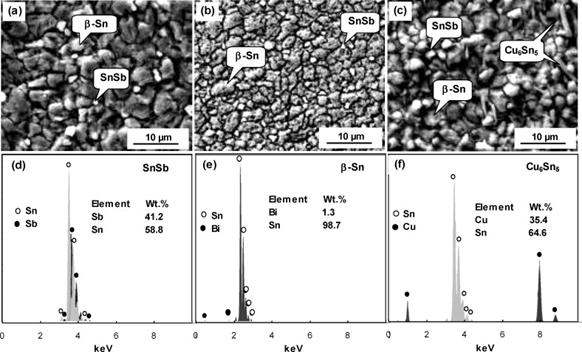
Figure 2, shows micrographs of the as cast sample (sp3), sample aged for 1hr (3-1), sample aged for 2hrs (3-2), sample aged for 3hrs (3-3), and sample aged for 4hrs (3-4). The micrograph of the as cast sample, labelled sp3 shows the inter-metallic compound phase appearing as white. The inter-metallic compound phase is SbSn, and it precipitates from the grain boundaries of the eutectic matrix. The matrix itself reveals a eutectic structure of two solid solutions- one tin-rich and the other lead-rich. According to Khanna [2], these properties of hardness, toughness and ductility cannot be found to the required degree in a single–phase alloy. Thus, inter-metallic compounds are hard and have a low coefficient of friction but are extremely brittle, whilst pure metals and solid solutions though ductile are usually soft and with a relatively high coefficient of friction. A suitable combination of mechanical properties can, however, is obtained by using an alloy in which particles of hard inter-metallic compounds are embedded in a matrix of ductile solid solution or, in some cases a eutectic of two solid solutions. Tin and antimony forms completely, soluble solution with lead in the liquid state, however, they are partly soluble in each other in the solid state. This is clearly shown in the microstructures which are in figure 2 which shows cored-β within some of the grains of the samples. Previous work carried out on similar or related alloys using Scanning Electron Microscope have clearly identified the presence of SbSn inter-metallic compound and β solid solution in the alloy see figure 3 and 4 [9-11].
The microstructure also shows particles which are dispersed within the matrix of the structure, these particles are seen in various quantities and distribution patterns as one views figure 2 (3-1), (3-2), (3-3), (3-4). The age- hardening of the samples must have affected the distribution pattern of the reinforcing particles (SiO2), this can be seen in figure 2 (3-4) where another phase segregated out and the hardness also dropped to 30 HRB. This may also be responsible for the changes observed in the hardness values of the samples at different ageing times. This fact has been confirmed by several researchers [12-16]. The distribution of the precipitated phase at the grain boundaries is not so much affected by the ageing process. It must however be admitted that it is more concentrated and segregated in the as cast state than the age-hardened samples. This could also be responsible for the variation in hardness values, some researchers have attested to the nature of distributed precipitates affecting the hardness values of alloys and composites [12-16].
|
|
|
|
(a) Cast sample (sp3) |
(b) Sample aged for 1hr (3-1) |
|
|
|
|
(c) Sample aged for 2hrs (3-2) |
(d) Sample aged for 3hrs (3-3) |
|
|
|
|
(e) Sample aged for 4hrs (3-4) (etched using 2% Nital solution) |
|
|
Figure 2. Micrographs x 1000 |
|
|
|
|
|
|
(a) Sn–5Sb |
(b) Sn–5Sb–3.5Ag |
(c) Sn–5Sb–1.5Au |
|
Figure 3. Microstructure of as-cast solder alloys |
||
|
|
|
Figure 4. SEM micrographs of: (a) Sn–5Sb, (b) Sn–5Sb–1.5Bi, (c) Sn–5Sb–1.5Cu, and EDX analyses of (d) SnSb, (e) β-Sn, and (f) Cu6Sn5 particles in the respective alloys |
Conclusions
The reinforcement (SiO2) has improved the hardness of the bearing alloy from 27.7 HRB to 33 HRB in the as cast state. The ageing-hardening of the composite led to a decrease in the hardness at certain ageing time before increasing again however at all the ageing temperatures the hardness was more than that of the bearing alloy alone.
The study has clearly shown that silica particles can be used in improving the hardness of 75Pb-15Sb-10Sn white bearing alloy metal and the morphology of the microstructure of the composite also agrees with the hardness values determined. The hardened 75Pb-15Sb-10Sn alloy can be used as bearing material for heavy duty bearings.
References
1. Higgins R.A., Properties of Engineering Materials, Fifth Edition, Hodder and Stoughton, London, 1985, p. 140-142.
2. Khanna O.P., Material Science and Metallurgy, 4th Reprint Dhanpat Rai Publications, 2008, p. 9-5.
3. Ihom A.P., Nyior G.B., Ibrahim G.Z., The Effect of thermal Ageing on Microstructure and some Mechanical properties of Al/2.0% Glass Reinforced composite, International J. Research and Reviews in Applied Sciences, 2012, 12 (3), p. 414-419.
4. Ihom A.P., Nyior G.B., Suleiman M., Ogbodo J., Precipitation hardening characteristics of Aluminum matrix-glass reinforced composite, Novus Scientia Journals, 2012, 1(3), p. 10-18.
5. Quartz, Microsoft Encarta, [DVD], Redmond, WA: Microsoft Corporation, 2008.
6. Ihom A.P., Nyior G.B., Anbua E.E., Ogbodo J.N., The Effect of Ageing Time on some Mechanical Properties of Aluminium/0.5% Glass Reinforced Particulate Composite, J. Of Minerals and Materials Characterization and Engineering, 2012, 11(9), p. 919-923.
7. Ashby M.F., Jones D.R.H., Engineering Materials 2, 27th Reprint Pergamon publishers Oxford, 2001 p. 93-97.
8. Bolton W., Materials for Engineering, 8th Reprint Butterworth-Heinemann Oxford, Great Britain, 2000, p. 86-87.
9. Ihom A.P., Hardness Improvement of Pb75%-Sb15%-Sn10% Bearing alloy through Reinforcement with 5% v/v SiO2 Particles, Usak University Journal of Material Sciences, 2012, 2, p. 205-212.
10. Esfandyarpour M.J., Mahmudi R., Microstructure and Tensile Behaviour of Sn-5Sb Lead-free Solder Alloy Containing Bi and Cu, J. Material Science and Engineering, 2011, 530, p. 402-410.
11. El-Daly A.A., Swilem Y., Hammad A.E., Creep properties of Sn–Sb based lead-free solder alloys, Journal of Alloys and Compounds, 2009, 471,(1–2), p. 98–104.
12. Taylor J.L., Basic Metallurgy for Nondestructive Testing, Revised Edition, The British Institute of Non-Destructive Testing, Cardiff, UK, 2000, p. 46.
13. Mathews F.L., Rawlings R.D., Composite Materials: Engineering and Science, 5th Edition, Woodhead Publishing Limited, Cambridge, England, 2005, p. 6-15.
14. Hassan S.B., Aponbiede O., Aigbodion V.S., Precipitation Hardening Characteristics of Al-Si-Fe/SiC Particulate Composites, Journal of Alloys and Compounds, 2007, p. 1-10.
15. Hassan S.B., Aigbodion, V.S., The Effect of Thermal Ageing on Microstructure and Mechanical Properties of Al-Si-Fe/ Mg alloys, Elsevier Journal of Alloys and Compounds, 2009, 486 (1-2), p. 309-314.
16. Onche O.E., The Development of Al-Cu Matrix Composite Reinforced with Diatomite, Ph.D Research Work FUT Minna Nigeria, 2009, p.1-10.